Despite advances in the fight against cancer, the disease still accounts for one in six deaths worldwide, with almost 10 million deaths in 2020. Current treatments such as radiation and chemotherapy have greatly extended patients’ lives and made some tumours survivable. But there are still types of cancer that are resistant to therapy. As a result, progress in reducing mortality is slow, while the global population is growing larger and older, resulting in an estimated 47% rise in the number of diagnosed cases by 2040 compared to 2020. The hope that the number of deaths will not follow the same trend depends on the likelihood that we will see some ground-breaking progress in treatments. Here are some of the promising approaches that could lead to breakthroughs in the coming years, using tools such as viruses, bacteria or cells from our own immune system, all with the help of genetic technologies.
VIRUSES THAT MAKE CANCER SICK
More than half a century ago, some doctors began noticing something unusual in their patients. After becoming infected with, for example, the influenza virus or the erysipelas bacteria, some cancer patients experienced inexplicable improvements. These curious cases kept appearing in which tumours sometimes disappeared completely and patients remained healthy for years or even decades. Various medical teams tried to treat cancer using natural viruses or bacteria, but overall it was a failure and the technique remained a dead-end.
Advances in science have revealed that infections have a significant, if complicated, link to cancer. Some viruses are capable of causing cancer (called oncogenic viruses), while others attack tumour cells (oncolytic viruses), and still others are capable of indirectly interfering with the development of the tumour through interaction with the immune system.
In recent decades, numerous research projects have sought to use genetically modified oncolytic viruses as potential treatments. In Spain, a team of researchers is testing this strategy using an adenovirus, the natural version of which causes conjunctivitis and cold symptoms. The virus, called VCN-01, has been modified into an oncolytic virus. The virus has shown efficacy in cellular models of paediatric tumours and has been used successfully to treat retinal cancer in a child.
“This virus is unprecedented and is much more powerful than the others that have been tested,” says Ramon Alemany, a researcher at the Translational Research Laboratory of the Catalan Institute of Oncology IDIBELL and co-founder of the biotech company VCN Biosciences, created to develop the project. The virus is designed to attack the tumour stroma, the scaffolding on which cancer cells are supported. When this collapses, the tumour cells “fall off and die,” explains Alemany.
VCN-01 is currently in clinical trials against pancreatic and head and neck cancers, with promising indications. Although it has not yet become a reality as a treatment, there is much hope for this approach. One such treatment has been approved for use in China since 2005 and several in the West are in the final stages of development before reaching the market.
The big problem with these therapies is that they don’t work the same in all patients and it’s impossible to predict in which patients they will be effective. This uncertainty about “treating ten patients to cure two,” as Alemany puts it, means that the cost-benefit ratio may not be attractive to Western pharmaceutical companies. Alemany says that the only thing that can be done is to keep testing. “With a little luck, in our case the cost-benefit may be worth it,” he says.
BACTERIA: FROM YOGHURT TO TUMOURS
Some researchers are exploring the possibility of using harmless bacteria to selectively target cancer, an avenue that had fallen by the wayside after repeated failures. The first attempts to treat cancer with bacterial infections date back to the mid-19th century. At that time, a cocktail of dead bacteria achieved extraordinary cures, such as that of a US boy who went from being on the brink of death from an abdominal tumour to surviving 26 more years cancer-free. But those treatments were unpredictable, worked only in some patients with no clear pattern, and the results were not reproducible, so the strategy was abandoned as the era of radiotherapy and chemotherapy dawned. Other attempts have experienced a similar fate, and only recently has some interest in the field reawakened, explains John Mark Tangney, a researcher at the Cork Cancer Research Centre in Ireland.
“Now our goal is to use bacteria as factories to produce whatever drug inside the tumor,” he explains. A few years ago, attempts were made to fight tumours with harmful bacteria such as salmonella, but this has so far failed to deliver the desired results. Tangney points out that the current trend is to use “good bacteria,” such as those used to produce yoghurt and other dairy products in the food industry or the bifidobacteria that already inhabit our intestines. “Almost any type of bacteria can live inside a tumour,” he says.

Oncolytic bacteria could attack tumours that are more difficult to eradicate. When they enter the body, these microbes seek out oxygen-deprived areas to proliferate. Interestingly, these areas are common inside tumours, beyond the blood vessels that keep the cancer alive. Chemotherapy and radiation rarely reach this deep into the tumour, so sometimes these internal areas are difficult to kill, allowing the cancer to re-emerge after the treatment finishes.
Tangney’s approach involves using genetically modified bacteria to produce a certain compound in the tumour. The patient is then given a prodrug, an inactivated drug that does not harm healthy cells. On contact with the enzymes produced by the bacteria, the drug is activated and selectively kills the tumour. Tangney’s team has achieved promising results in mice using bacteria that produce tumour necrosis factor alpha, a molecule that the body generates itself as a mediator of chemical messages between cells. “One of my goals is to promote the concept of good bacteria, because many of them are definitely beneficial to us,” he says.
WEAPONS FOR THE IMMUNE SYSTEM
In December 2012, the case of Emily Whitehead gained worldwide attention. The six-year-old girl was suffering from acute lymphoblastic leukaemia, the most common cancer in children, which is lethal if left untreated. Today Emily is alive and still cancer-free, nine years after undergoing experimental treatment. She is living proof that the immune system can be taught to fight tumours. In general, cancer cells are analogous to spy planes that are not detected by radar. They do this by producing proteins that turn off the attack of our defences and allow the tumour to grow unthreatened.
Whitehead was included in a clinical trial focusing on adult patients with chronic leukaemia who had relapsed several times after conventional treatments. The procedure involves removing millions of the patient’s T-lymphocytes, immune system cells responsible for activating defences against infection. Researchers use genetic technologies—initially viral vehicles were used, but today the trend is to use the gene-editing tool CRISPR—to introduce into these cells a molecule capable of recognising the tumour and attacking it; this molecule is called chimeric antigen receptor, which gives these cells the name CAR-T. They are then reintroduced into the patient and left to do their work. It is therefore an immunotherapy, as it is based on arming the immune system to fight the cancer, either with the patient’s own T-cells or from a donor.
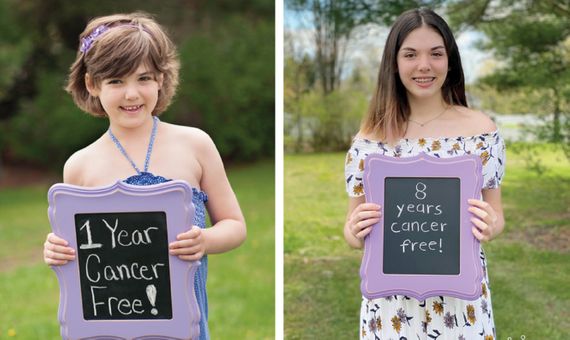
CAR-T cell therapy is only one of the possible alternatives in cancer immunotherapy, although it is the one that has experienced the greatest growth in recent years. Several are already approved for leukaemias and lymphomas, although they have yet to prove their usefulness in solid tumours. Potential long-term adverse effects also need to be studied, as there are risks of neuronal toxicity and uncontrolled activation of the immune system. However, the researchers are optimistic. “I think this strategy will have less toxicity and will also mean that if the tumour comes back, the patient’s own body will be able to recognise and reject it. This is the future,” says Marta Alonso, a researcher at the Centre for Applied Medical Research (CIMA) at the University Hospital of Navarra, who is studying the use of viruses to treat another of the tumours with the worst prognosis in adults and children: brain glioblastoma
CORRECTING ERRORS IN CANCER CELLS
Another of the most innovative ways to attack cancer is through epigenetics. This term refers to chemical changes that occur in genes that don’t alter the DNA sequence, but function as switches, turning them on or off. Many of these changes are key to the proper functioning of the body, but others are faulty and can cause various diseases, including cancer. Epigenetics is strongly implicated in the action of environmental carcinogens. But just as epigenetic errors can lead to cancer, correcting them can help to cure it.

According to Manel Esteller, one of the leading experts in cancer epigenetics in Spain who works at the Bellvitge Biomedical Research Institute, there are already several epigenetic cancer drugs on the market that are being used to treat leukaemia and myeloma. Some of these drugs target types of cancer for which until now there were only palliative treatments, he points out. In the near future, the genetics of each patient will also be used to predict their response to different drugs so as to administer the most appropriate one.
Comments on this publication