Among the research currently being developed on COVID-19 are those that analyze the possibility that a transposon or transposable element within the gene ACE1 (Angiotensin-Converting Enzyme 1 ), and not the ACE2 enzyme that has been talked about so much until now, may be protecting people who carry this transposon against the effects of SARS-CoV-2.

ACE enzymes
In the current pandemic, the important role played by one of the virus’s entry proteins into cells is already clear. It’s the ACE2 protein present on the surface of cells, although it also runs free in our bloodstream. This protein is an enzyme that has a protective effect on blood pressure as it acts on a vasoconstrictor protein, turning it into a vasodilator. But also, since they are in the cell membrane, some of its areas serve as a receptor for the coronavirus that penetrates through the cells. And based on the latter, much research is being done on whether the higher or lower production of ACE2 may be the basis of a higher or lower susceptibility to the virus and the higher or lower infectivity of the virus. Thus, there is research that suggests that the greater morbidity against the virus in men compared to women may be due to higher levels of ACE2 in men.
But the ACE2 protein is part of a complex system —the renin-angiotensin system— where various enzymes are involved, and where various products that act on the heart, kidney, brain, immune system, etc. originate from. In particular, the production of the enzyme ACE2 is closely linked to that of the enzyme ACE1, given this name because it was the first to be discovered. In fact, ACE1 acts in the system, passing a component, angiotensin I, which is not functional, to angiotensin II, which is a vasoconstrictor that raises blood pressure, and can cause damage to various organs such as the lungs or the heart. ACE2 subsequently acts on angiotensin II, converting it to angiotensin 1-7, which is a vasodilator that lowers blood pressure and avoids its adverse effects.
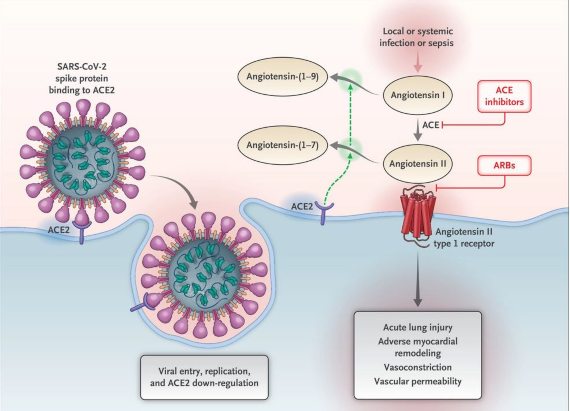
Interaction between SARS-CoV-2 and the Renin-Angiotensin-Aldosterone system
Here we see the initial entry of coronavirus 2, severe acute respiratory syndrome (SARS-CoV-2) into cells, mainly type II pneumocytes, after binding to its functional receptor, the angiotensin converting enzyme 2 (ACE2). Following the endocytosis of the viral complex, the cell surface enzyme ACE2 would decline in function, resulting in an unopposed accumulation of angiotensin II. Local activation of the renin-angiotensin-aldosterone system may mediate lung injury responses to viral aggressions. The boxes in red (ACE inhibitors and ARBs that are Angiotensin receptor blockers) are the pharmaceutical products that are used to block the enzyme ACE (1), the first, and the second, ARBs, to block the angiotensin II receptors that exist in cell membranes.
With this enzymatic chain, the proper functioning of the body system depends on the coordinated production of both enzymes and a good ACE1 / ACE2 balance, so that anything that may affect this relationship can have harmful effects on our health. In the case of the coronavirus, since the virus is complexed with ACE2, this may cause the amount of angiotensin II to increase in infected people as it cannot be transferred to the angiotensin 1-7 because the virus is “sequestered” by the virus, the ACE2 enzyme that converts the first into the second. Remember that the first is a vasoconstrictor and the second a vasodilator, and that all this in the end would result in hypertension and more risk in the evolution of people infected by the coronavirus.
This is why in the case of virus infection, not only is the amount of ACE2 enzyme produced by the body important, so is the amount of ACE11 enzyme that is produced. And this is where the transposon existing inside the Ace1 gene comes into play.
An interesting transposon of ACE1
Inserted into the human genome, as in the rest of living beings, there are a multitude of transposons whose size range from several thousand pairs of nucleotides to a few hundred. The latter is the case of the transposon that occurs in the ACE1 enzyme gene. It is a transposon with about 300 pairs of nucleotides called Alu, because it has a restriction site for an enzyme called just that because it is obtained from the bacterium Arthrobacter luteus (Alu). There are millions of Alu sequences in the human genome. Many of these sequences are inserted in their corresponding positions in all human genomes. Those Alus are said to be fixed in the genome. But there are some Alu sequences that are present in only some people´s genes. In this case it is said that they are polymorphic Alu.

In the ACE1 gene in particular there is a polymorphic Alu sequence inserted towards the end of the gene as it is transcribed into RNA. This gene has several thousand pairs of nucleotides. There are people who carry this Alu in their two Ace1 genes: they are homozygous I / I, I insertion of Alu. Others that carry it in one of the genes: they are heterozygous, I / D, D deletion. Finally, there are other people for whom the transposon is absent in the two genes: they are homozygous D/D.
When this Alu in present is some people, especially in homozygous, I / I, a lower level of synthesis of this enzyme tends to occur, which in the end points to a lower amount of the vasoconstrictor agent, angiotensin II, in their bodies. For this reason, people with this Alu sequence would present fewer problems with hypertension, which in turn may have a protective effect on their developing COVID-19.
The polymorphic Alu of ACE1 and the coronavirus
Two different studies suggest that the presence of this Alu in the ACE1 gene may have a certain protective effect on the development of COVID-19.
Firstly, these studies in human populations measure and compare the frequency with which this Alu sequence occurs and the number of infected or killed by the coronavirus. Studies such as those by Yamamoto et al. (2020) and Delanghe et al. (2020)2 show that human populations with the lowest incidence of COVID-19 are those of East Asia, which are precisely those in which the frequency of the polymorphic Alu genotype I/I is higher. On the other hand, genotype I/I is less frequent in Central Europe, Mediterranean Europe and the U.S. so that infections and deaths from the coronavirus are greater. According to these findings, the East Asian populations could have a certain genetic protection factor against the effects of the virus due to the higher frequency of this transposon.

In addition, there are studies where an analysis is made of the development of the disease in infected people depending on whether they are carriers or not of Alu. This was the case of the study carried out in Spain by Gómez et al. (2020) 3 in which 204 infected patients (67 severe in ICU) and 536 age-matched control groups were analyzed. The study found that people with the D / D genotype for the Alu—that is, they don´t carry it in any of their Ace1 genes—tend to have more serious infections compared to those that carry it in their two genes, especially if they are hypertensive men.
This initial data suggests that the presence of the polymorphic Alu existing in the Ace1 gene can serve as a form of protection against the effects of SARS-CoV-2 in some people. Especially when it is homozygous, that is, when Alu is present in the two Ace1 genes that each person carries. Naturally, more controlled population-based cohort studies are needed to confirm this possible protection. But if confirmed, it would have to be taken into account when treating infected people.
BIBLIOGRAPHY
1. Pagliaro, P. & C. Penna. 2020. ACE/ACE2 Ratio: A key also in 2019 Coronavirus Disease (Covid-19)? Frontiers in Medicine. 7: 335.
2. Delanghe, J.R. et al.2020. COVID-19 infections are also affected by human ACE1 D/I polymorphism. Clin. Chem. Lab. Med. 25; 58(7):1125-1126.
3. Gómez, J. et al. 2020. Angiotensin-converting enzymes (ACE, ACE2) gene variants and COVID-19 outcome.
Manuel Ruiz Rejón and
Fernando Ruiz Rejón,
Retired cardiologist from the Ramón y Cajal Hospital in Madrid
Comments on this publication