Sixty years ago, British geneticist Mary Frances Lyon (1925-2014) proposed a hypothesis stating that a specific inactivation process occurs in women’s sex chromosomes — and those of female mammals in general — transforming them into cellular mosaics. This hypothesis, which has been known ever since as lyonization, has been proven fully valid in our times, having led to interesting advances and discoveries.

LYONIZATION
At the end of the 1950s, it was discovered that the pair of sex chromosomes in males is comprised of a normally sized chromosome, the X chromosome inherited from the mother, and a very small chromosome, the Y, inherited from the father. In women, however, this pair consists of two X chromosomes, one from the father and one from the mother. It was then deduced that women have double the X chromosome genes than men, which is no small matter given that the X chromosome contains more than a thousand genes, whereas the Y has only about 75. Thus arose the enigma: are genes from both X chromosomes expressed within the female’s cells or is there some kind of compensation for women’s double dose of genes (compared to males).
Faced with this dilemma, Lyon first proposed that one of the somatic cell’s two X chromosomes in women — and in female mammals, in general — is inactivated in the phase when genes are expressed, in interphase1. Specifically what happens is that the inactivated X chromosome is completely condensed — a process that is called heterochromatinization — whereas the other X chromosome and the rest of the chromosomes are decondensed and expressed in RNA and proteins. The inactive X chromosome would be the origin of the sex chromatin or Barr body, a more darkly stained body observed in the nucleus of a female cell and which is absent in male cells. Even though female cells have a double dose of X-linked genes, the process of inactivation caused by heterochromatinization results in only one of the set — the one that has not been converted into heterochromatin — being active, thus compensating for the double dose in females, as compared to males who only have one X.

Secondly, Lyon proposed that inactivation occurs in the first phases of female development — in fact just two short hours from fertilization — and at random: in some cells the X chromosome inherited from the father is inactivated and in others it is the X chromosome from the mother. Thirdly, she sustained that once a specific X chromosome has been inactivated in a specific embryonic cell, all of the cells that originate from it will inherit said inactivation, with the result that all of the cells in the area of the adult body that this particular cell gave rise to will be inactive. This explains why organisms with coloration patches in the fur are usually female and not male. This is the case with tabby cats with black and orange patches on a white background. These cats would be heterozygous for the gene that determines the colors orange and black, which is located in the X chromosomes. The patches of color reflect the zones that have developed from cells that have inactivated one or the other of the X chromosomes. This is why in some zones one of the color alleles — the orange — is expressed and in other zones the other allele — the black — is expressed.
Therefore, and in conclusion, what has been known as lyonization causes mammalian females to be mosaics, where the genes of one or the other of the two X chromosomes carried in the cells are expressed, a phenomenon that does not happen in males because their cells contain only genes of a single X chromosome, in addition to the Y
REVOLUTIONARY GENETIC CONCEPTS RELATED TO LYONIZATION
Although Mary Lyon’s hypothesis about the inner workings of women’s (and mammalian females’) X chromosomes was at first met with some reservations, in later years it was generally accepted. Particularly because much of the research that her hypothesis prompted (some of it even conducted by her) received convincing genetic and cytologic results which supported the theory. Once confirmed, it represented the starting point for several discoveries, some of interest for theoretical biology and others with implications for clinical genetics.
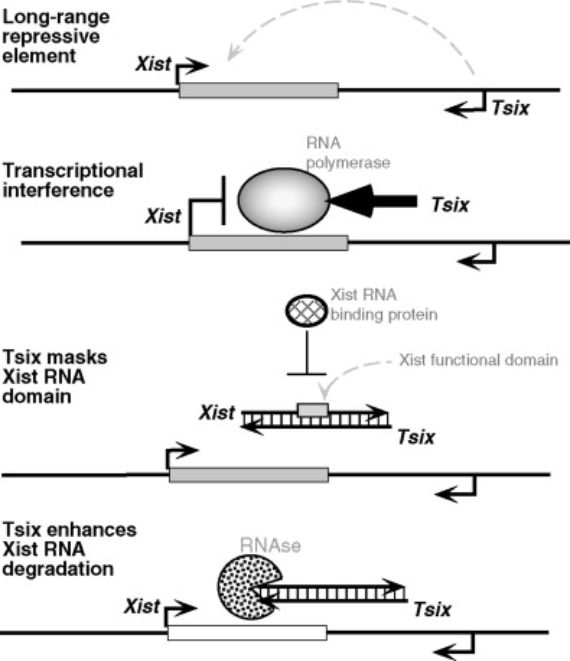
Thirty years after Lyon’s initial work on the process, it was discovered2 that the chromosome X-inactivation in females is controlled by the very X chromosome to be inactivated. And as opposed to what normally happens, this is not because a protein comes from some other chromosome or from the same X chromosome. It happens because in the X chromosome where a gene is going to be inactivated, the Xist (for “X inactive specific transcript”) gene synthesizes a long RNA — it has more than 15,000 ribonucleotides — that in binding itself along the chromosome subjects the chromosome to the heterochromatinization process, inactivating it, and thus preventing the transcription process that occurs first with the messenger RNA and later with proteins. This was one of the first times that it was found that a long non-coding RNA (a non-protein coding transcript) could have a gene activation regulatory function, something that was later seen to occur in various biological situations.
Another exceptional genetic mechanism has been found to be related to female active X chromosomes. Specifically what happens in this chromosome is that another RNA (different from the previously mentioned one) is synthesized from the Xist gene. But this happens from the opposite strand of the gene’s double helix — and in the opposite direction — from that which is transcribed in the inactive chromosome. This RNA is called Tsix, Xist backwards. It is even longer than the Xist; it has 40,000 bases, and with its synthesis in the active X chromosome, it prevents the synthesis of the Xist RNA with the inactivating function3. It is also one of the first cases in which it was observed that two different RNAs were transcribed from a single gene: one in one direction of the gene’s double helix and another in the other direction, and where, in addition, one of the RNAs interferes with the other: if the Tsix impedes the Xist synthesis, it would be an anti-Xist.
CLINICAL IMPLICATIONS OF LYONIZATION
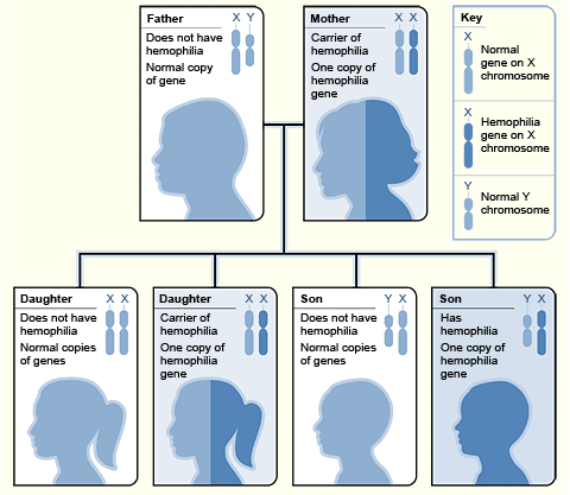
The phenomenon of lyonization that occurs during the development of women and mammalian females in general could also have clinical ramifications. Women are generally less likely than men to suffer diseases caused by gene mutations of the X chromosome. This is due to the fact that women have two X chromosomes, and if one carries a mutated gene, it can be substitute with the normal gene that comes from the other X chromosome. This does not occur in males because they only have one X chromosome. This is what happens with diseases like hemophilia, which involves hemorrhaging caused by a lack of a coagulation factors. In this case, men can be hemophiliacs due to a genetic mutation of their single X chromosome from which these factors are synthesized. In contrast, although women can be carriers of the mutated gene in one of their X chromosomes, with the normal gene in the other X, enough coagulation proteins are produced to prevent hemorrhaging, event though it is only expressed in half of her cells and that only fifty percent of the normal coagulation factors are produced. This level is adequate to protect them from hemorrhaging, especially because the coagulation factors are released from the cells and circulate through the bloodstream where they can act upon the whole body.
But there are other situations in which the presence of an X chromosome with a functioning gene opposite a mutated gene in the other X chromosome of women does not protect them from the disease. This is what occurs, for example, in Rett syndrome, which is mainly prevalent in women with symptoms that include a serious cognitive disability — a severe form of autism — that becomes increasingly worse throughout the patient’s life4. This syndrome occurs as a result of a mutation in a gene in the X chromosome from which a protein is synthesized. The protein binds to the DNA in various locations and regulates the expression of many other genes. This occurs most of all in the brain. Whereas women with the mutation in both X chromosome genes or men with the mutation in their single X chromosome would not be viable, heterozygous (with one normal and one mutated gene) women are viable, but suffer from this syndrome. In this case what happens is that only half the brain cells of the patients express the normal gene — the cells that have inactivated the mutated X. But the other half, having inactivated the X with the normal gene, does not produce the protein. Half of the nerve cell production of the protein is not adequate for normal brain function.
PROSPECTS AND CONCLUSION
As we have said, the lyonization hypothesis has continued to prove valid through the present day. Though it continues to be a field of active research in many respects. First of all, much about the inactivation mechanism itself is being investigated: research on all the protein factors that interact with the RNA that react first, research on the effects of these factors on the DNA structure and the genes that are inactivated, etc. Secondly, there is ongoing research on the possible deviations in the process: there are cases in which inactivation does not occur randomly, but rather in some women activation occurs with a preference for either the paternal or maternal X chromosome; and research on the existence of genes that escape inactivation in the X chromosome that is heterochromatinized. All this research is also of clinical interest, for example, the latter two phenomena can play a role in certain illnesses and kinds of cancer. Likewise, very recently there has been investigation into to what degree men’s greater sensitivity to the coronavirus (SARS-CoV-19) could be related to their greater production of the cell membrane protein, ACE2 (Angio-tensin Converting Enzyme 2), which the virus uses to enter MRC-5 cells. The gene that controls this enzyme is located in the X chromosome, and if what happens is that men produce greater amounts of ACE2 than women and are therefore more susceptible to infections, the question is: How is this difference produced when Lyon’s hypothesis predicts that there should be equal production between the two sexes. Could this difference be due to the behavior of male hormones on the gene?
The pioneer behind this entire field of research, Mary F. Lyon, saw her hypothesis accepted and her work recognized as she was invited to join such important scientific societies such as the Royal Society (although she met with opposition) or the National Academy of Sciences in the U.S. She even lived to see her her name on a laboratory in the institute where she developed her work: the Mary Lyon Centre in Harwell, and in 2014 the establishment of the Mary Lyon Medal from the U.K. Genetics Society. However, she was not awarded the Nobel Prize, which many scientists claim she deserved for her revolutionary, comprehensive, and productive hypothesis.
Bibliography
- Lyon, MF. 1961.Gene Action in the X-chromosome of the mouse. Nature, 190:372-373. The first in a set of works in which the hypothesis is developed.
- Brown, C.J. et al.1991. A gene from the region of the human X inactive centre is expressed exclusively from the Inactive X chromosome. Nature, 349: 38-44.
- Lee JT et al.1999. Tsix, a gene antisense to Xist at the X inactivation center. Nat Genet, 21(4):400-4.
- Bienvenu, T. et al. 2000. MECP2 mutation account for most cases of typical forms of Rett syndrome. Hum. Mol. Genet. 9:1377-1384
- Sama, I.E et al. 14 May-2020. Circulating plasma concentration of Angiotensin Converting Enzyme 2 in men and women. Eur.Heart Journal. 41(19): 1810-1817.
Comments on this publication