In 2009, Hans Clevers of Utrecht University in the Netherlands succeeded in creating microscopic structures from stem cells that simulate the villi of the small intestine. These so-called “organoids” are 3D cell cultures that mimic the structure and function of real organs, allowing the study of organ development, function and disease. But one type deserves its own chapter: brain organoids, which raise ethical dilemmas that don’t exist for a liver or a kidney.
Organoids are an example of collaborative science with multiple contributors. Attempts to grow tissues or regenerate organs from animals in vitro have been made since the early 20th century, but it was only at the turn of the 21st century that stem cell technologies and new three-dimensional cell culture matrices gave the field a boost. Before Clevers, David Odde created liver organoids in 2006, and the progress has been spectacular. Organoids offer more faithful biological models of humans than animals and could one day lead to replacement organs, the dream of personalised medicine.
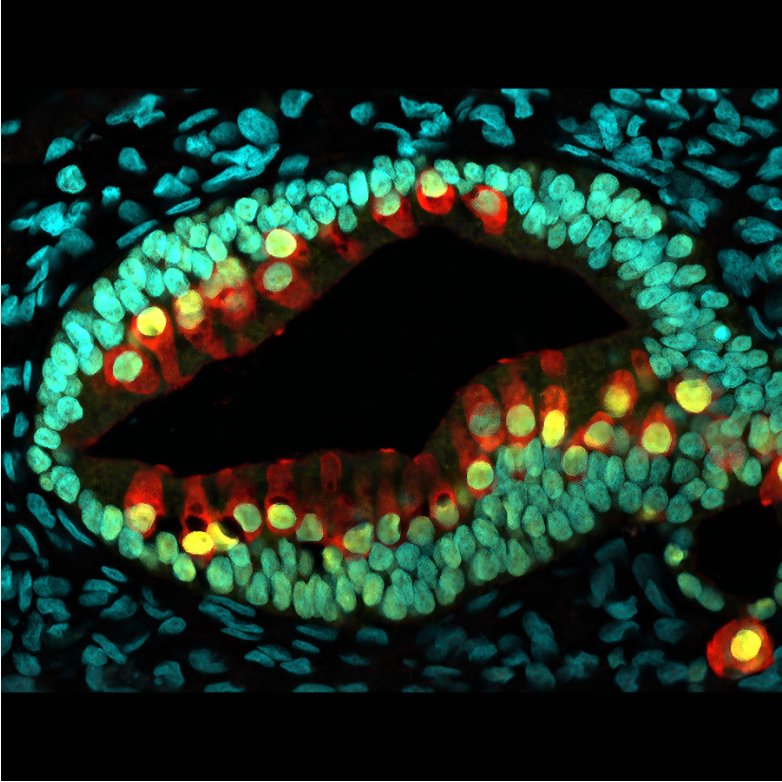
Brain organoids are particularly interesting because the brains of mice or rats cannot simulate the complexity of the human brain, and these mini-brains may reveal crucial clues for the study of neurodegenerative diseases such as Alzheimer’s or conditions such as autism spectrum disorder.
In 2008, at the Riken Institute in Kobe, Japan, Yoshiki Sasai created tissue cultures of cerebral cortex, the first rudimentary brain organoids. Five years later, at the Austrian Academy of Sciences, Jürgen Knoblich and Madeline Lancaster were making pea-sized brain organoids that simulated the organisation of an embryonic brain. At the time, Knoblich warned journalist Ed Yong in The Scientist that the organoids his team had created were not “brains-in-a-jar”, and that they were a far cry from an adult brain. But in some respects that gap has narrowed.
The possibility of consciousness
One of the technical limitations in creating organoids is that of size, because if they get too big, the cells inside are deprived of nutrients. But human brain organoids have now been made to produce their own blood vessels, and have been successfully transplanted into the brains of mice and rats, so that the cells remain alive and responsive to stimuli such as light or images. Brain organoids that develop rudimentary eye structures on their own have also been created.
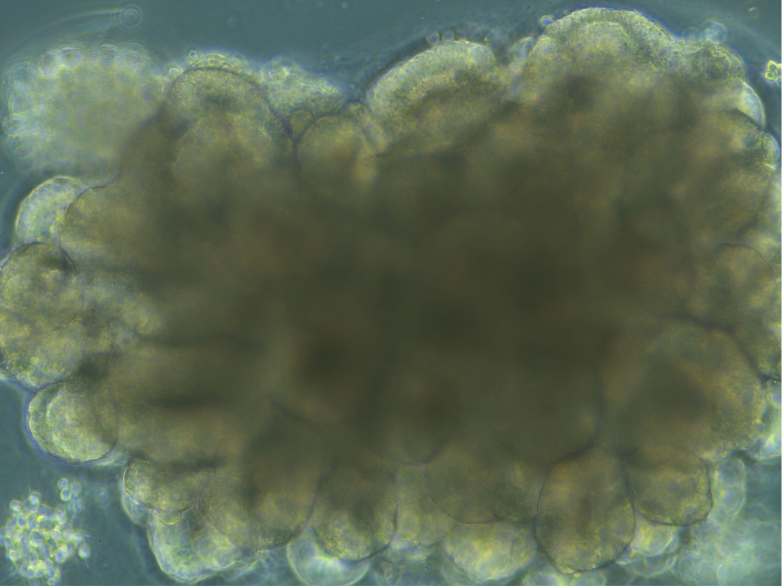
In 2019, Lancaster managed to get a human mini-brain made up of about two million neurons—the adult brain has about 86 billion—similar to a 12- or 13-week-old foetal brain, to spontaneously connect to muscle fibres or the spinal cord of mice, as neurons do during development, and this connection was able to induce muscle contraction. The researcher explained to The Guardian that these organoids were still too primitive to have anything resembling thoughts, feelings or consciousness, but that the discussion should continue.
Also in 2019, at the University of California, Alysson Muotri observed electrochemical activity in cortical organoid neurons that bore some resemblance to that of the brain of a premature baby. The organoids are not whole brains, but tiny cell masses representing only the cerebral cortex, and the researchers manipulated the neurons to deprive them of an essential protein. For Thomas Hartung, a researcher at Johns Hopkins University who also obtained electrical activity in brain organoids, this represents “a primitive kind of ‘thinking'”, albeit in a purely mechanical way.
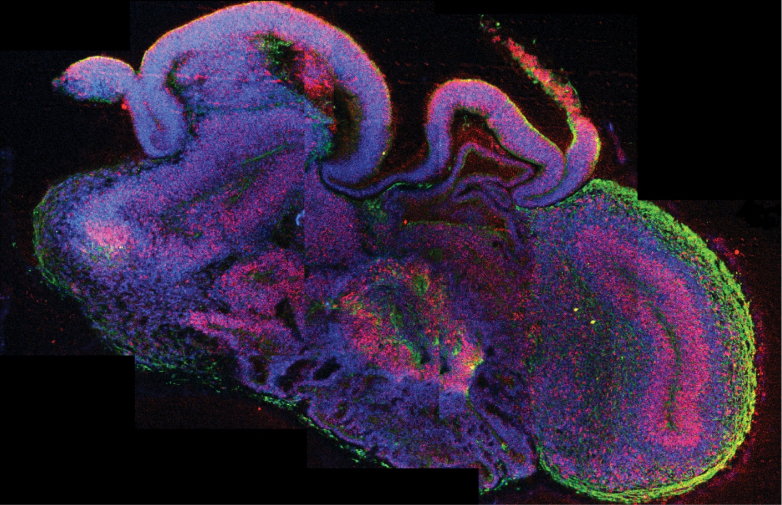
On the occasion of the presentation of Muotri’s work, neuroscientist Christof Koch of the Allen Institute for Brain Science warned in Nature: “The closer they get to the preterm infant, the more they should worry”. Bioethicists Julian Koplin and Julian Savulescu of the University of Melbourne wrote that such research is morally justified for the medical benefits it can bring, but that there are ethical issues not covered by current stem cell regulation. According to the two experts, the possibility of consciousness would put an organoid not on a par with a human being, but with animals that can suffer and feel pain.
The legal status of the organoids
The problem, University of Pennsylvania neuroscientist Hongjun Song told OpenMind, is that “we do not have a clear idea what consciousness is”. Koplin and Savalescu suggested that we should “restrict research with brain organoids that resemble the brains of fetuses beyond 20 weeks’ gestational age—the earliest estimate for when consciousness develops in human beings,” and that, in any case, there should be a presumption that consciousness might exist before assuming otherwise.

The urgency for ethical guidelines intensifies with each new development. Song commented that organoids could eventually function as biological computers that “can be trained to perform simple tasks”, and this prediction is beginning to come true: in 2022, Brett Kagan of the Australian company Cortical Labs connected organoid brains to a computer system and taught them to play Pong, a tennis video game from the 1970s. Kagan argued that his mini-brains are the first “sentient” ones, as they are “responsive to sensory impressions through adaptive internal processes”. Other experts question this claim; but Hartung, Kagan, Muotri and others have already defined a new frontier of biocomputing that aspires to surpass Artificial Intelligence: “organoid intelligence“.
For all these reasons, voices such as Newcastle University law expert Joshua Jowitt have been raising the possibility that brain organoids might qualify for the legal status of “person”, which is not the same as “human”: companies are legal persons. But researchers in Japan and Taiwan led by Tsutomu Sawai of Hiroshima University go further, suggesting that in the future they may deserve to be considered natural persons—that is, humans. “This issue will soon become urgent once brain organoid technology has been further developed,” says Sawai. “In preparation for that time, it is essential to examine the accompanying questions thoroughly and in advance; we have taken the first step in that direction.”
Comments on this publication