With the growing social awareness that the world inhabited by human beings does not have infinite resources to exploit, there are increasing incentives for research into new ways of recycling products that have previously been considered waste, in some cases replacing raw materials, which can then be put to other uses.
An example of the latter can be found in the manufacture of bioplastics from vegetal waste. In the field of alternatives to plastic it is important not to be misled, since not all biodegradable plastics dispense with petroleum as a raw material, and not all bioplastics—those that do—are biodegradable. With the intention of achieving the dual objective of eliminating dependence on oil in the manufacture of plastics and also improving their biodegradability, the chemical industry has been developing new compounds derived from vegetal matter. Starch, cellulose and other biopolymers are used as a starting point in the chemical processes that will result in bioplastics, and these base substances are obtained from corn, peas, potatoes, cotton, hemp or wood. However, this approach has a serious drawback in terms of sustainability, as it requires the diversion of raw materials that can be used for other purposes, which in turn increases the land area devoted to cultivating these crops.
This is why researchers are looking to use plant waste that normally has no other use. At the Italian Institute of Technology in Genoa (Italy), Athanassia Athanassiou and Ilker Bayer have developed a method for manufacturing bioplastics from plant waste such as rice husks, cocoa pods, spinach or parsley stalks and carrot or cauliflower refuse. The origin of the idea stems from a modification of the method for producing cellophane from cotton and hemp cellulose, which led to the discovery of how a mouldable plastic mass could be obtained by dissolving the plant matter in trifluoroacetic acid, without all the treatment normally used.
The researchers tried using other alternative sources of cellulose and found that the material had different properties depending on the source material. Thus, the bioplastic obtained from spinach stalk was more flexible than the one derived from rice, for example. In this way, the scientists suggest, there may be a new use for the parts “we don’t want to eat,” although much research is still needed to turn this technology into an economically viable substitute for the use of refined vegetal products.
A further twist on the use of waste to make bioplastics can be found in the US company Newlight Technologies, whose starting material is a greenhouse gas: methane produced by the flatulence of cattle on farms or by the decomposition of organic matter in covered landfills. They call it AirCarbon and it has already been used to make chairs, mobile phone cases and Dell computer casings. The technology in this case involves taking advantage of the carbon present in the methane and combining it in a series of chemical processes, thanks to a biocatalyst developed by Newlight, to form a thermoplastic, 60% of which is composed of elements from the methane and 40% of oxygen from the air. According to the manufacturers, the properties are the same as those of petroleum-based plastic.
But despite the above, all plastic always has a downside—sooner or later it tends to be discarded as waste. 91% of plastic is never recycled, and experts often warn that biodegradable plastic does not break down as quickly as we have been led to believe, so it accumulates for years. Therefore, another possibility is to convert plastic waste into a distinct material with a different use—for example, fuel. This is the aim of chemical recycling, a method that involves degrading the long polymer chains into smaller fragments through gasification or pyrolysis processes. In this way, fuels such as diesel or kerosene, or mixtures similar to crude oil, can be obtained.

Another option is to give plastics a definitive use, and for this there is probably no better idea than to build cities with them, as children do with their block toys. Researchers such as Sibele Cestari of Queen’s University Belfast, together with several companies, are working on transforming recycled plastics into bricks, tiles, beams and other building elements. According to Cestari, crucial milestones have already been achieved in both the mechanical properties of these materials and their aesthetic qualities, and while the technology is not yet ready for large-scale application, progress is very promising.
Recycled obsolescence
While technology can provide new forms of recycling, technological advances can also make objects now in use become obsolete and start piling up in landfills. A glaring case in point is that of car batteries, which are currently recycled. 90% of the lead recovered from the processing of spent batteries is used to manufacture new batteries of the same type. However, the automotive industry is moving in the direction of replacing these lead-acid batteries with more efficient lithium-ion batteries, making the previously recycled lead a potential environmental pollutant.
Angela Belcher from the Massachusetts Institute of Technology (USA) estimates that more than 200 million of these lead-acid batteries will pile up in the United States alone. And it will be partly her fault, since her research group is dedicated to developing better and more efficient lithium batteries, including through genetic engineering to create nanowires with scaffolding formed by the assembly of bacteriophage viruses. Fortunately, the researcher is also working on finding a way to use this lead from old batteries for the manufacture of solar panels.
A new experimental type of photovoltaic cell uses a lead compound with a crystalline structure similar to that of a mineral called perovskite as a material that absorbs light rays. This material is undergoing further development in terms of improved efficiency, and has already surpassed established technologies based on solar cells that use thin films of silicon. Besides their efficiency, the main advantage of perovskite-structured solar cells is their low manufacturing cost, but they have the drawback of requiring lead, a highly contaminating metal whose extraction is also harmful to the environment.
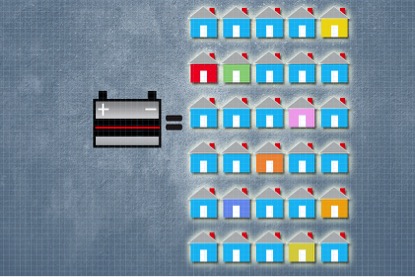
Belcher’s work shows that it is feasible to use lead from used car batteries to manufacture this type of solar cell. According to her analysis, lead from a single battery can be used to make enough solar panels to power 30 homes. And when these solar panels need to be replaced at the end of their useful life, the lead they contain can be recycled again.
An increasingly pressing need for technological recycling is that of electronic devices. The continuous advance of consumer technologies means that immense quantities of so-called e-waste, or electronic waste, are generated every year, and this is now considered the fastest growing type of waste. In 2019, 53.6 million tons of this waste were generated worldwide—an average of 7.3 kilos per person—and it is estimated that by 2030 the figure will increase to 74.7 million tons.
Among this waste are large quantities of precious metals. According to Sandra Wilson at the University of Dundee, 1 gram of gold, the amount contained in many wedding rings, can be recovered from about 41 mobile phones. At Coventry University, Sebastien Farnaud is researching the use of bacteria to recover metals from electronic waste, something that is applied in mining to extract the minerals from the ore. This avoids the dumping of these metals into the environment and also reduces reliance on mining.

In addition, devices such as smartphones, wind turbines and electric car batteries, among other devices, make use of so-called rare earths, elements that are not necessarily scarce in the earth’s crust but are widely distributed and difficult to find in their pure state and to extract. Mobile phones contain about 60 different elements, including rare earths such as yttrium, lanthanum, terbium, neodymium, gadolinium and praseodymium. Only about one-fifth of global e-waste is recycled, so huge amounts of these materials are being wasted.
In 2019, Apple began using recycled rare earths in a component of its iPhones for the first time. But there is still a long way to go before recycling becomes an industry standard, as the procedures for recovering rare earths from devices are energetically costly and polluting. Researchers such as Cristina Pozo-Gonzalo, from Deakin University (Australia), are working on alternative methods such as electroplating to recover these materials using cleaner and more sustainable techniques. Thus, thanks to increased recycling and advances in research, the waste we generate could be the raw materials mine of the future.
Comments on this publication