On 7 January 2022, American David Bennett became the first human to receive a transplanted heart from a pig genetically modified to minimise rejection. The procedure was successfully completed at the University of Maryland Medical Center under the direction of cardiothoracic surgeon Bartley Griffith. Unfortunately, Bennett only lived for two months with his new heart, but the pioneering experience has given impetus to the field of xenotransplantation, the use of animal organs as an alternative to address the shortage of donations.
Xenotransplantation represents one of the latest breakthroughs in organ replacement, although it is not a new idea. The first failed attempts date back to the early 20th century, when the basics of immune rejection were not even known. Since then, several such transplants have been performed, albeit with limited survival times, along with countless pre-clinical animal trials.
In September 2021, surgeons at NYU Langone Health transplanted the first kidney from a genetically modified pig into a human body—for research purposes only, as the patient was clinically dead. The pigs used for both this procedure and Bennett’s came from the company Revivicor, and have several genetic modifications including the removal of certain enzymes responsible for the production of a type of sugar that causes immune rejection in humans.
However, xenotransplantation not only still has technical limitations, but is also subject to ethical debate, both in terms of the recipients—Bennett was deemed ineligible for a human transplant, but his status as an ex-convict was also discussed—and the use of animals for this purpose.
Improvements in organ preservation by perfusion
But while this avenue is progressing, the field of allotransplantation—the usual human-to-human organ transplantation—has also seen progress in recent years. One of the most notable advances is the use of perfusion machines. The method generally used is static cold storage, in which the organ is kept refrigerated and at rest, with its metabolism slowed down, until transplantation: the classic icebox. However, in recent years, the technique of circulating a mixture of oxygen and nutrients through the blood vessels of the organ to preserve it, either at low temperature (hypothermic perfusion) or body temperature (normothermic perfusion), has grown in recent years.
Specifically, normothermic or warm perfusion is the closest thing to keeping the organ functioning normally while outside the body, as it preserves the active metabolism. This technique, which extends the life of tissues, broadens the range of donors, makes it possible to use organs in conditions that would preclude them from being transplanted with static storage, and even allows the administration of drugs to, for example, dissolve a blood clot.
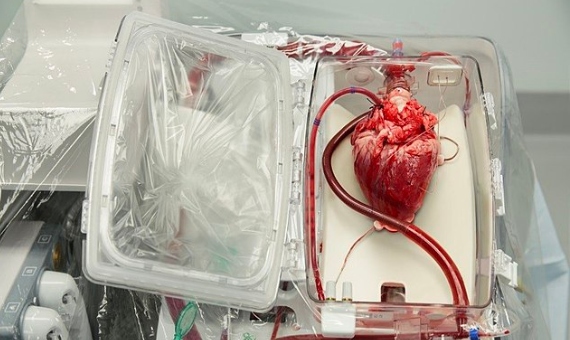
Perfusion was first used for kidneys, but has now been extended to heart, liver, lungs or pancreas. One example is the Organ Care System (OCS), an invention of a Massachusetts company TransMedics, which revives the donated heart and keeps it at body temperature, nourished and oxygenated, while it waits to be transplanted. The creators of the new technique believe it will increase the rate of successful heart transplants by 30%.
OCS has also been used for liver and lung transplantation. “Lungs are very sensitive and can easily be damaged during the donation process. The cold-storage method does not allow for reconditioning of the lungs before transplantation, but this promising ‘breathing-lung’ technology enables us to potentially improve the function of the donor lungs before they are placed in the recipient,” explains Abbas Ardehali, a cardiothoracic surgeon at the University of California, Los Angeles. When Joseph Murray performed the first successful organ transplant—a kidney—in 1954, he had no idea how far we would go with these surgical techniques.
Pioneers in the transplantation of new organs
Other important milestones in this field have been reached in recent years. In 2014, a Swedish woman became the first to give birth with a transplanted uterus, received from an anonymous 61-year-old donor. By April 2021, a review counted at least 23 cases of women giving birth with transplanted wombs. Another reproductive organ was at the centre of the challenge faced by Curtis L. Cetrulo and his team of surgeons at Massachusetts General Hospital in Boston in early 2016 when they attempted to transplant a complete penis. The 15-hour procedure, which involved reconstructing complex vascular and neural structures so that the organ fully retained its urinary and sexual function, was a success. It was the first in the USA, but two similar operations had already been carried out in China and South Africa, and several more have been performed since.
On 27 March 2010, surgeon Juan Pere Barret and his thirty or so collaborators also left the operating theatre of the Vall d’Hebron University Hospital in Barcelona triumphant after completing the world’s first full face transplant. The first partial face transplant was achieved in France in 2005, and in 2008 a team from the Cleveland Clinic in Ohio performed the first near-total human face and upper jaw transplant, which allowed their patient, a 46-year-old woman, to even regain the ability to speak and the senses of smell and taste. Since then, several dozen such operations have been performed, in some cases combined with limb transplantation: in 2020, a transplant of the face and both hands was performed at NYU Langone Health.

At the University of Texas (USA), another important page in the history of surgery was written in early 2016 when Dr. Jesse C. Selber and his colleagues performed a multiple transplant of the skull, scalp, kidney and pancreas. This operation involved a combination of many different tissues, such as skin, muscle, blood vessels, nerves and bone, but also complete organs.
Stem cells and 3D printing, the future of transplantation
A likely insurmountable barrier to whole organ transplantation is the brain, a field that has been much talked about in the media in recent years but is generally regarded by experts as science refiction. But this does not mean that transplantation technologies have no place in the fight against pathologies that damage brain tissue, such as Alzheimer’s or Parkinson’s disease. In particular, the use of stem cells is seen as a way forward for the regeneration of damaged tissues, including the nervous system.
Scientists at University of Tel Aviv were able to replicate a functional heart using 3D printing techniques in 2019. Video: BBC News
Stem cells are also the foundation for the creation of organoids and the bioengineering of tissues and organs. In the first case, this involves creating tiny versions of organs in vitro that could in the future provide sources for transplants, and which are currently proving invaluable for research and drug testing. As for bioengineering, it is moving towards the manufacture of complete organs using 3D bioprinting systems. In 2019, Tal Dvir and his collaborators at Tel Aviv University created the first complete human heart—albeit on a small scale—3D bioprinted from a patient’s cells and biological materials. In the future, it may even be possible to transplant bioengineered penises, as Anthony Atala has successfully tested on rabbits at the Wake Forest Institute for Regenerative Medicine in North Carolina.
For organ rejection problems, which require minimising the activity of the recipient’s immune system—with the risk of disease—scientists are exploring new strategies. In recent years, experimentation has begun with a gene therapy that programmes certain lymphocytes (called CAR-Treg, or chimeric antigen receptor regulatory T-cells) to defend the new organ from attack by our own immune system, which normally tries to destroy it when it detects a foreign element. Perfusion machines are also being used to acclimatise the organ to the recipient’s immune system so that rejection is reduced. These are brilliant ideas aimed at making the majority of transplants successful.
Comments on this publication