1997 was the year of cloning. With the announcement in February of that year of the birth of the world’s first successfully cloned mammal—Dolly the sheep—actually born the previous year, cloning became not only the hot scientific topic of the day, but also one of the most topical, endlessly talked about and debated in the media and the public arena because of its potential application to humans. This interest continued during the early years of the current century. But a quarter of a century after Dolly, cloning has disappeared from the spotlight without the dystopian predictions of cloned humans or the great hopes of these techniques for biomedicine having materialised.
Dolly was not the first animal cloned, nor the first mammal, nor the first sheep, but she was the first mammal—not the first animal—cloned from an adult cell. The technique, called somatic cell nuclear transfer (SCNT), involves replacing the nucleus of an egg cell, which contains the DNA, with that of an adult cell from another individual. The embryo generated, and thus the resulting individual, is genetically identical in its nuclear DNA to the adult cell donor. This “Dolly method” has been regularly used in animal cloning to copy specimens of interest. In 2018, Chinese researchers led by Qiang Sung of the Chinese Academy of Sciences published a study in Cell about the obtention of the first monkeys cloned by SCNT, named Zhong Zhong and Hua.
Cloning to reproduce organs and tissues
Such reproductive cloning in humans is prohibited in many countries, although some do allow therapeutic cloning. The principle of the technique is the same, but its purpose is different; in this case, the aim is not to produce a cloned individual, but to use the embryo generated to derive stem cell lines to create replacement organs and tissues that are genetically compatible with the donor, as well as for research and drug testing.
In 2004 and 2005, the Korean researcher Hwang Woo-suk published two studies in the journal Science in which he claimed to have obtained human embryonic stem cells by cloning, but his experiments were shown to be fraudulent. In 2013, a team led by Shoukhrat Mitalipov at the Oregon Health & Science University confirmed the first human embryonic stem cells obtained by SCNT from foetal skin cells. The following year, two teams generated embryonic stem cell lines by SCNT from adult human cells.
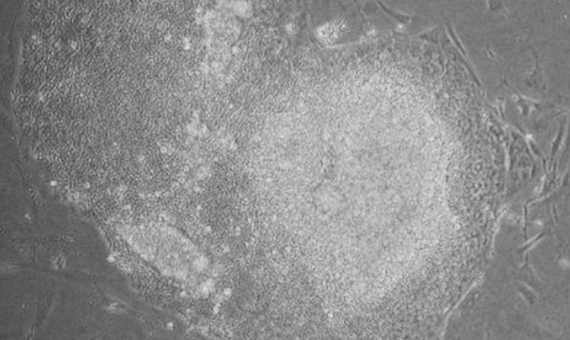
However, the use of embryos to generate stem cells is the subject of ethical debate and has been rejected by broad sectors of society. In 2006, science found a possible alternative when a team led by Shinya Yamanaka of Kyoto University succeeded in reprogramming adult cells to obtain stem cell lines, known as induced pluripotent stem cells (iPSCs). The work earned Yamanaka the Nobel Prize in Physiology or Medicine in 2012.
iPSCs and embryonic cells are not completely equivalent, and both techniques have pros and cons, beyond the fact that the controversy over embryos has shifted the work of numerous researchers and project funding toward iPSCs. As for human cloning, while it is theoretically possible today, it is a very inefficient process. At present, neither therapeutic cloning nor the iPSC method has yet managed to overcome all the technical obstacles that would allow these technologies to move beyond experimental trials towards widespread use in regenerative medicine. In 2021, skin cells were reprogrammed for the first time to create embryonic-like structures, adding another tool to this growing field of research, which, however, has not yet begun to realise its therapeutic promises.
Comments on this publication